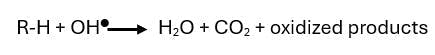What is advanced oxidation process for wastewater treatment?
Key Highlights
- Ozone-based advanced oxidation, radiation-driven, catalytic, and AOPs involving physical methods and high energy reactions such as thermal activation are the four categories of AOP.
- Chemicals containing oxygen radicals such as hydroxyl radicals and chlorinated compounds are commonly used in AOP.
- The water solubility of the chemical to be treated is the basic requirement of AOP because the process occurs in water.
- The formation of the hydroxyl radical or any other reactive oxygen species is the first and the most important step in the treatment of the water-soluble pollutant in AOP.
- Advantages of AOP include the ability to directly degrade volatile, water-soluble organic pollutants such as methyl tert-butyl ether (MTBE), to disinfect the water due to the use of UV, and no requirement for microbial involvement.
Imagine that the wastewater is contaminated with methyl tert-butyl ether (MTBE), a water-soluble gasoline additive. It spilled on the ground, washed off into the water, and it eventually landed in the wastewater. Suppose we need to find a way to directly remove MTBE in the wastewater without the use of biological processes while also disinfecting the water. This is where advanced oxidation process (AOP) is useful.
AOP is an in-situ treatment to degrade contaminants in wastewater using highly reactive radicals. It should not be confused with the conventional oxidation process which involves the AOP process among other steps such as sedimentation, pH adjustment, and post-filtration. The effectiveness of AOP in wastewater treatment is due to the dynamic yet straightforward chemical reactions that provide advantages to AOP yet also some disadvantages.
Categories of AOPs
There are four categories of AOPs as follows:
- Ozone-based advanced oxidation: These AOPs involve ozonation and peroxone. Peroxone is a process that involves combining ozone and hydrogen peroxide.
- Radiation-driven: These could be homogenous AOPs or heterogenous photocatalytic AOPs. They involve the use of ozone along with chemicals such as chlorine (Cl2), chlorine dioxide (ClO2), persulfate, peroxide, and vacuum ultraviolet (VUV) light.
- Catalytic: Catalytic AOP involves Fenton reactions. In these reactions, ferrous iron catalyzes hydrogen peroxide to produce hydroxyl radicals.
- Other AOPs: These involve physical methods and high energy reactions. Examples include the use of ultrasound, plasma, thermal activation of persulfate, and electrochemical oxidation.
Notably, some of the chemical reactions can overlap in the above categories. For example, UV or ozonation can be applied to both ozone-based oxidation and radiation-driven AOPs. Another example: photo-fenton reactions can be applied in both radiation-driven AOPs and catalytic AOPs.
What chemicals are used in AOP?
Chemicals containing reactive oxygen species are typically used in AOP. Specifically, the list includes the following:
- Hydroxyl radicals (●OH) and reactive oxygen species;
- Radicals derived from persulfate, peroxide, chlorinated compounds, carbonate, and nitrate;
- Dissolved inorganic constituents; and
- Solvated electrons.
Other chemicals containing dissolved inorganic constituents and solvated electrons are also used. These chemicals affect process kinetics, reaction mechanisms, and product formation during the degradation of contaminants in the wastewater.
Basic requirements of AOP
The main requirement of AOP is that the chemical that needed for removal must be soluble in water because AOP involves chemical oxidation reactions that occur in the water. As such, AOP is limited to treating water soluble chemicals.
How does AOP remove water-soluble pollutants?
In general, the water-soluble pollutant in the water undergoes oxidative degradation due to the presence of highly reactive radicals, the most common being the hydroxyl radical (OH●). This results in the degradation of the pollutant. The general chemical reaction for AOP is as follows:
Where:
- R-H: water-soluble pollutant
- •OH: hydroxyl radical
The specific chemistry involving the removal of pollutants via AOP can be illustrated in three steps as follows:
- The formation of the hydroxyl radical
- The initial attack by the hydroxyl radical on the target pollutant
- Subsequent attacks by the hydroxyl radical until the pollutant is mineralized.
The mineralized product settles down in the tank and can be removed by sedimentation or filtration.
As step 1 shows, the hydroxyl radical must be formed first before the target pollutant can be degraded in steps 2 and 3. The mechanism for ●OH production depends on the category of the AOP used.
Examples of how the hydroxyl radicals are formed
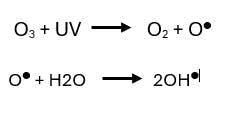
Ozone-based AOP
In the ozone based AOP method, the reaction of UV with ozone (O3) produces hydroxyl radicals (●OH) as follows:
Photocatalytic oxidation
In photocatalytic oxidation, titanium dioxide (TiO2) is typically used as the photocatalytic surface. When UV light falls on the surface of TiO2, electrons are produced and on the surface, they participate in the redox reactions to produce superoxide radical anion and hydroxyl radicals.
What are the advantages of AOP?
AOP relies on chemical oxidation reactions involving reactive oxygen species, commonly with hydroxyl radicals. These reactions can completely oxidize organic pollutants into carbon dioxide and water. They also offer several advantages to AOP as follows:
- There is no need for biological processes. Because AOP is a chemical reaction, there is no need for microorganisms that are involved in biodegradation. Here, reactive oxygen species such as the hydroxyl radicals are involved in chemical reactions involving oxidation.
- Ability to directly degrade volatile, water-soluble organic pollutants. Volatile organic pollutants such as MTBE could contaminate wastewater. To treat water contaminated with MTBE, MTBE needs to be physically transferred to a gas phase using a technique called air stripping or to the solid phase using adsorption followed by additional processes such as thermal or catalytic oxidation. In contrast, the use of AOP to remove MTBE from wastewater is a simpler process. Because MTBE is soluble in water, AOP degrades MTBE directly in water through chemical reactions.
- Disinfection ability. The two categories of AOP, namely ozone-based advanced oxidation and radiation-driven reactions involve the use of ozone and peroxide. While these are used to induce the formation of reactive oxygen species, they are also disinfectants because they remove microorganisms from the water.
Where is AOP commonly applied?
AOP is commonly applied to wastewater containing water-soluble chemicals. One example of where AOP is applied is for the treatment of landfill leachate in wastewater. Leachate is formed on a landfill when the solid content in a landfill mixes with the water. It then travels into other sources such as the underlying soil or to the nearby water bodies. As such, it can contaminate the wastewater. Example of pollutants in wastewater originating from landfill leachate are dissolved organic matter and xenobiotic organic compounds and these could be degraded by AOP if they are water soluble.
Factors to consider before using AOP
There are several factors to consider before AOP, a green technology, can be applied to wastewater, but the big three are cost, appropriate technology and the ability to remove residual hydrogen peroxide.
As for cost, the chemical reagents required for the chemical oxidation reactions in AOP such as peroxide or titanium dioxide can be expensive. Much like other chemical costs associated with wastewater treatment, this chemical expense must be considered.
Selecting the appropriate AOP category for your application is also critical. There are four categories of AOP discussed above and wastewater operators need to select the category that will meet their treatment needs. Similarly to the chemical expenses, there are different costs associated with the different categories of AOP technologies to consider.
Finally, consider also the wastewater plants' ability to remove residual hydrogen peroxide. Wastewater treatment that uses hydrogen peroxide in AOP need to monitor for the presence of residuals, which can be harmful. Carefully designing and using the AOP treatment can prevent excess residual hydrogen peroxide in the system, but this should also be considered in addition to the cost and technology choice.
Wastewater treatment plants that are looking to treat water soluble organic contaminants such as MTBE should consider the above factors to decide if AOP is the best treatment route. As is the case with other technologies, AOP can be used in combination with other treatment methods notably electrocoagulation and filtration. However, it should be emphasized that costs, compatibility with the wastewater treatment plant, and treating residual pollutants are among the factors that should be considered prior to treatment implementation.
About the Author
Saleha Kuzniewski
Saleha Kuzniewski, Ph.D. has authored several publications in the fields of scientific research, biotechnology, and environmental regulations. She is the winner of the 2023 Apex award for publication excellence. She is also the founder of Environmental Remediation & Innovations, LLC. Kuzniewski can be reached at [email protected].