Many Uses of Microsensors
Figure 1. Various types of microsensors for biofilm and corrosion studies: dissolved oxygen microsensor, cobalt-based phosphate detection microsensor, biofilm application, pH measurement at a corroded iron coupon, 3-D microprofiling set up, and needle-type micro electromechanical system microsensor
The development of electrochemical microsensors in micro-scale is a revolutionary advancement for non-destructive and in situ measurements of both absolute levels and changes in chemical and biological species in engineered and natural aquatic systems. A needle-type electrochemical microsensor represents one of the most prominent methods for better understanding fundamental mechanisms contributing to the rates and magnitude of liquid-solid (e.g., corrosion and biofilm) interfacial chemical and biological reactions (see Figure 1, page 17).
The needle-type microsensor is able to penetrate soft samples (e.g., biofilm, sediments and biological tissues) and is capable of probing the full depth of a model system to determine the profile of relevant water quality parameters and enable calculations of the relative contribution of related biological and chemical reactions.
In using microsensors, various kinetic parameters, including net specific consumption, production rates, fluxes, and diffusion coefficient can be estimated from the measured concentration profiles. These include pH, redox potential, ammonia, nitrate, nitrite, free chlorine (or monochloramine), phosphate and dissolved oxygen (DO) concentration microprofiles. Biofilm thickness, diffusion boundary layer (DBL) and stratification of chemical and biological reactions also can be determined.
Corrosion Mechanism Investigations
Corrosion and biocorrosion in drinking water distribution systems can cause serious economic, environmental and public safety problems from pipe damage, water loss and water quality degradation. Corrosion studies have been conducted for decades; however, most of them have focused on bulk concentration monitoring and metal surface characterization.
Although the methods used have provided important information, they are indirect and/or forensic investigation and do not provide direct information. Microsensors have been utilized to collect in situ micro-spatiotemporal water chemistry data near metal surfaces (approximately 10 µ) with regard to long-term corrosion processes under various water conditions (see Figure 2, page 18). By measuring pH, phosphate as a corrosion inhibitor, lead, DO, redox potential, free chlorine (or monochloramine), the corrosion mechanisms at the metal surface have been further elucidated. This confirms what others have previously speculated or indicated by alternate “macro” methods. Better understanding of corrosion, like lead release from premise plumbing, can lead to better corrosion control strategies to prevent water quality non-compliant events.
Figure 2. 2-D pH contour map near metal service (approximately 10 µ) of 14 days aged brass/lead solder joint coupon with flow (2 mL per minute) versus understagnation. Water conditions were pH 9, 100 mg/L Cl-, 100 mg/L SO42-, 10 mg/L C DIC, and 4 mg/L Cl2 free chlorine.
Emulsion Stability Characterizations
Bilgewater is a regulated mixture of seawater and any water contaminated with oils, solvents, surfactants and particulate matter. Free or suspended oils (mechanical emulsion) can be readily separated from the water phase by simple physical processes like skimming. However, chemical oil-in-water emulsions formed in bilgewater are notoriously difficult to treat, which may result in non-compliance with existing ocean discharge regulations.
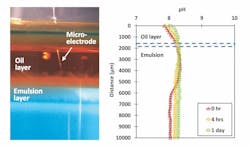
Unlike mechanical emulsions, chemical emulsions tend to remain in a stable homogeneous state for an indefinite period of time. It is critical to develop innovative methods for breaking emulsions to ensure onboard, oily liquid waste treatment meets discharge standards. For effective prevention and treatment solutions, fundamental knowledge concerning the formation, occurrence and behaviors of shipboard oil-in-water emulsions needs to be addressed. Microsensor techniques have been used for characterizing emulsion stability and chemical properties (see Figure 3, page 20).
By combining with a variety of characterization methods, including zeta potential, contact angle, interfacial tension and confocal laser scanning microscopy analysis, the formation and stabilization of oil-in-water emulsions become better understood. A recent microsensor emulsion study found that anionic surfactant stabilized emulsions are affected more by salinity than nonionic surfactant emulsions. This led to development of a strategy for effective bilgewater management in shipboard applications.
Water Monitoring Microsensors
Chlorine is the disinfectant most frequently used for drinking water and wastewater treatment to remove unwanted organisms. Approximately 40% of the ballast treatment systems tested for International Maritime Organization Type Approval incorporate chlorination, and almost all use neutralization to reduce total residual oxidation prior to discharge.
Residual chlorine control is imperative for ships that employ oxidants for treatment. Accurate and reliable process control would allow for the optimization of power and chemicals while meeting the discharge standards. Given that an affordable and reliable online residual chlorine sensor is not currently available commercially, microsensors can provide an opportunity for online process control with minimal calibration requirements and reliable measurements.
One example is use of microfabrication and miniaturization technologies, which can offer several advantages, including reduced manufacturing cost, increased integration and potentially reduced power consumption due to smaller size. The integration of multiple microsensors into a single device kit that is replaceable and disposable after several measurements is a good example for field applications with low maintenance.
Figure 3. Oil-in-water stability tests using pH microsensors. The left image shows a photograph of a microsensor application and the right shows spatial and temporal pH changes in oil-in-water emulsions (1% standard oil mixture) with anionic surfactant. The point at which the sensor signal was obtained by contact with the oil is represented by 0 µ.
Heavy Metal Monitoring Microsensors
To estimate the detrimental role of heavy metals in various natural media, it is important to determine the concentration of heavy metals (e.g., arsenic, lead, chromium and cadmium) in water, soil and groundwater. Because of mercury’s toxicity, mercury-based electrodes have been replaced with bismuth electrodes.
Bismuth (Bi) is an environmentally friendly element with low toxicity and widespread pharmaceutical use. Generally, in Bi-based analytical systems, Bi(III) ions (0.25 to 1 ppm) are directly added into the sample solution and then simultaneously deposited on the gold or carbon substrate along with target heavy metals. However, in some cases, this method is not applicable for onsite heavy metal measurement in field applications because it introduces additional Bi ions.
One way to overcome this problem is the development of a thin-film chip microsensor, which is able to rapidly measure heavy metals with small sample volumes. The use of a Bi-electroplated thin-film chip microsensor for measuring heavy metals seems quite promising. It has many advantages over conventional methods, including a simple structure, high-sensing performance, and compatibility with other advanced microfabrication technologies.
The Bi-coated film electrode has received considerable attention due to its low toxicity when compared with mercury, advantageous electrochemical properties, and insensitivity to DO. It is widely accepted that square-wave anodic stripping voltammetry is one of the most sensitive, convenient, and cost-effective analytical methods for detection and determination of heavy metals in aqueous solutions.
The application of electrochemical microsensors has high potential for collaborations between disciplines in material science, oceanology, biology, chemistry, mechanical engineering, medicine and agriculture. Utilizing microsensor techniques can directly measure chemical or biological species at high spatial resolutions, which may provide more clear evidence that has not been observed through conventional means.
Electrochemical microsensors have mainly been used for characterizing biofilm in many environmental applications, such as drinking water distribution system, bio-membrane processes, wastewater treatment, bioremediation, and transport and fate of benthic contaminants in sediments.
The quantitative evaluation of biofilm-related microenvironments will assist in understanding the mechanics of the associated biological processes and lead to the improvement of kinetic modeling and process designing. For further extensive use of these microsensors beyond micro-scale analysis, with a modification of the developed microsensors (e.g., improvement of robustness, fouling prevention and field deployment), the microsensor technique can be applied for integrated water quality monitoring systems for in situ monitoring of chemical and biological reactions over a large area to monitor algal blooms or chemical and oil spill events.