Per- and polyfluoroalkyl substances (PFAS) are synthetic fluorinated organic compounds widely used in industrial, commercial and consumer products due to their oil and water repellent characteristics.
Product applications include oil and water repellent surface coatings for packaging, textiles and cookware (e.g., pizza boxes, fabric coating, and non-stick frying pans) and firefighting foams. Over 600 manufactured compounds can be classified as PFAS, including perfluorooctanoic acid (PFOA) and perfluorooctanyl sulfonate (PFOS). These two perfluorinated compounds were produced in the largest amounts within the U.S. but were largely phased out and replaced by shorter-chained analogs over the last 15 to 20 years.
The presence of PFAS in water supplies is concerning for affected water systems because of their potential for adverse health effects at low nanogram-per-liter (ng/L) concentrations and the limited removal achieved through conventional treatment processes. With this in mind, in May 2016, the U.S. EPA established a 70 ng/L drinking water health advisory (HA) for the sum of PFOA and PFOS. Since then, more than 10 states have promulgated enforceable standards, some of which are significantly lower than the federal HAs. Furthermore, many utilities have established their own goals to meet the federal HAs or more stringent goals, such as California’s Notification Levels of 6.5 ng/L for PFOS and 5.1 ng/L for PFOA. A number of states are also developing receiving water quality standards and limits in biosolids that could significantly affect water reclamation facilities in the future.
Meanwhile, a number of federal legislative efforts in 2019 were proposed to compel the U.S. EPA to regulate PFAS across all water-related media. As one of the first steps under its PFAS Action Plan, in February 2020, the USEPA announced its intent to develop enforceable MCLs for PFOS, PFOA and potentially, other PFAS compounds.
These regulatory developments have put water and wastewater agencies on edge and in some cases, left them scrambling to comply with rapidly changing requirements. To date, regulation of PFAS has affected mainly drinking water utilities, especially those relying on groundwater contaminated with PFAS. However, recognizing that typical wastewater effluent and biosolids contain background concentrations of PFAS in almost every region of the country, regulation of PFAS in discharges and biosolids could have much far-ranging impacts.
PFAS in Potable Reuse
Over the past few decades, potable reuse has emerged from obscurity and is now generally accepted as a potential source of new water supply. Potable reuse is a compelling water supply option in many water-stressed places because it is both local and drought resistant. Even parts of the U.S. not typically considered water-short are now pursuing potable reuse because it can provide the dual benefits of water supply and nutrient discharge avoidance. Still, potable reuse sometimes suffers from lingering issues with public perception. Given the recent impact of PFAS on other aspects of the water and wastewater industry, it is important to determine the consequences potential new regulation andnew public awareness of PFAS will have for potable reuse projects.
In conventional groundwater treatment projects designed to address PFAS, the treatment technology of choice is typically (pseudo-)adsorptive, namely granular activated carbon (GAC) or ion exchange. Other treatment methods are typically ineffective or cost-prohibitive. However, the calculus on PFAS treatment is different in potable reuse, in which cost- and energy-intensive processes are the norm. The vast majority of potable reuse projects will implement some form of advanced water treatment (AWT), which will rely on a small number of core treatment processes to remove both regulated and unregulated chemical constituents. These processes can be grouped roughly into two categories: membrane-based AWT and carbon-based AWT.
Membrane-Based AWT
Most existing potable reuse projects rely on reverse osmosis (RO) as the key treatment process for removing unwanted dissolved constituents. However, these membranes are extremely effective at removing PFAS compounds as well.
Figure 1 demonstrates this effectiveness at the Raw Water Production Facility Direct Potable Reuse (DPR) Facility in Big Spring, Texas (adapted from Steinle-Darling et al., 2016). In 2013, this facility became the first and only operating municipal DPR project in the U.S. There, treatment consists of microfiltration (MF), RO, and UV advanced oxidation prior to blending with other raw surface water supplies.
Many U.S. facilities use this treatment model for indirect potable reuse via groundwater recharge. The challenge with this approach is the concentrate produced by the RO process, which constitutes approximately 15% of flow. As concerns about PFAS compounds in the environment mount, their presence in RO concentrate may contribute to challenges with its disposal.
Carbon-Based AWT
Carbon-based AWT is a compelling alternative to the brine-producing membrane-based AWT, especially inland, where brine disposal is challenging. Typical carbon-based AWT applies a combination of ozone (O3), biologically active filtration (BAF) and GAC as central treatment units, supplemented by substantial additional pathogen inactivation or removal processes, such as UV, chlorine, MF, or ultrafilters (UF).
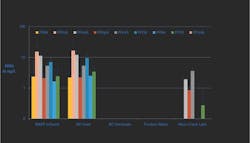
Figure 2 shows the removal of PFOA and PFOS through a non-membrane-based treatment process employed at permanent DPR demonstration facility. This facility employs the following treatment processes: O3, BAF, UF, GAC, and UV, with a future chlorination step planned for full-scale implementation. While the BAF and UF steps achieve some PFOS and PFOA removal (presumably due to removal of particle-associated PFAS compounds), GAC achieves the majority of the removal (to below detection).
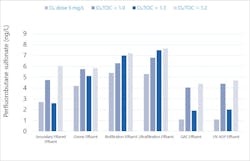
As shown in Figure 3, different and interesting behavior can be seen in the shorter-chained PFAS compounds, such as perfluorobutane sulfonate (PFBS), a 4-carbon acid. Keeping in mind that the graph’s scale spans less than 10 ng/L in total, concentrations progressively increase through treatment before being substantially removed by GAC. One working hypothesis for the increasing trend is that larger parent compounds are being degraded into PFBS through ozonation.
The GAC process in this treatment train addresses the TOC remaining after the other processes, both to meet the Florida potable reuse limit of 3 mg/L and to limit the downstream formation of
disinfection byproducts.
The GAC step is thus not simply a second biological filter; achieving the 3 mg/L TOC target consumes adsorptive capacity, requiring carbon change-out approximately every six months. This is pertinent, since a change-out was performed between the second and third sample events. Removal of PFBS was less effective with older carbon (second and fourth sample events) than with fresh carbon (first and third sample events).
Conclusions
The frenzied regulatory activity surrounding PFAS is necessitating a hard look at the fate of these compounds in all areas of water and wastewater treatment. Certainly, treatment designed specifically for the removal of PFAS will be necessary for many drinking water facilities and could be needed for some wastewater applications.
However, for potable reuse projects, the crisis is less acute: in many cases, concentrations in conventional recycled water will already be lower than regulatory limits for drinking, and where they are not, AWT processes already in place in typical potable reuse projects will address
them adequately.
Furthermore, it is more than happenstance that potable reuse projects are well-positioned to address the presence of PFAS. The potable reuse industry as a whole has been proactive in addressing a large range of constituents of emerging concern through extensive monitoring and advanced treatment systems designed to address not only regulated parameters but many others as well.
Thus, we have come to this new challenge with a large amount of data already in-hand, and with projects designed to remove not just this set of compounds-du-jour but many other future contaminants yet to be discovered as well.
About the author: Eva Steinle-Darling is reuse innovation lead and vice president for Carollo Engineers Inc. Steinle-Darling can be reached at [email protected].