Flat Sheet Membrane Bioreactors (MBR) Efficacy
Amos Branch is water reuse technologist, Nicola Fontaine is principal technologist and associate vice president, Andrew Gilmore is MBR chief technologist and vice president, and Andrew Salveson is water reuse practice director for Carollo Engineers. Larry Morris is R&D scientist, Hiro Kuge is technology manager and Siamak Modaressi is application engineer, and Yasushi Terao is general manager of the Kubota Water & Environment R&D Center for Kubota Membrane USA Corp. These authors can be reached at [email protected].
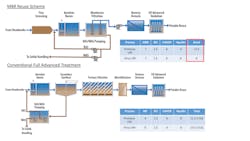
undefinedChallenge testing has demonstrated high protozoa and virus log reduction values (LRV) by membrane bioreactors (MBRs), greater than six in many cases. Nevertheless, accredited LRVs are often lower and sometimes zero. The State of California Division of Drinking Water (DDW) requires 12-log virus, 10-log Giardia, and 10-log Cryptosporidium removal across an indirect potable reuse (IPR) scheme (SWRCB, 2018) and other states (e.g., Nevada, Texas) have similar risk reduction-based goals for these pathogens. In order to replace conventional activated sludge, tertiary filtration and micro- or ultra-filtration (MF/UF), in a typical membrane based advanced water reuse scheme—i.e., MBR, reverse osmosis (RO), ultraviolet (UV) disinfection and advanced oxidation process (AOP)—an MBR would need to be accredited for more than 2.5 LRV for protozoa. Additional virus removal would be beneficial as it would allow for either less travel time or requirement for chlorination (see Figure 1).
MBRs remove pathogens by a combination of size exclusion by the membrane and biofilm layer, adsorption and wasting with waste-activated sludge and biological predation. The effectiveness and relative contribution of each mechanism to overall reduction is pathogen and system specific.
California is now considering a tiered validation protocol for MBRs in water reuse to provide methods for defining LRVs with MBRs. Work to develop the validation protocols is being done under the Water Research Foundation (WRF) Projects 4997 (Tier 1 and Tier 2) and Project 4959 (Tier 3). In the tiered system, conservative default LRVs based on existing data can be assigned for Tier 1 (1-log viruses, 2.5-log protozoa), detailed validation studies can be performed to demonstrate higher MBR product-specific LRVs (Tier 2), or a correlation can be established between surrogate parameters and LRVs (Tier 3). Tier 3 is more difficult due to the diverse mechanisms by which MBRs remove pathogens.
Kubota membranes used in MBRs are a flat sheet configuration and have an average and maximum pore size of 0.2 and 0.4 µm, respectively (see Figure 2).
The larger pore size relative to some other competing membrane products has resulted in a perception that overall LRV will be lower. In addition, the flat sheet configuration is not suitable for application of pressure decay testing at test pressures typically applied, due to the geometric arrangement of the module. The challenges with integrity monitoring and perceived impact of a larger pore size have made it difficult for the industry—in the absence of robust data—to assign LRVs to Kubota membrane products. Until this study, there is no data reporting Cryptosporidium and Giardia removal for Kubota membranes, and only a sparing set of indicator microorganism data when compared to hollow fiber competitors. Also, previous sampling efforts on Kubota systems (and others) suffered from poor analytical recoveries and low sample volumes, which reduced the LRV able to be demonstrated.
The goal of this study was to evaluate the pathogen removal performance for the purpose of validating Kubota membranes according to proposed Tier 2 validation requirements. The membranes at the plant sampled have been operated continuously for nearly 10 years without replacement and would be considered near the end of their typical usable life. Sampling after such a prolonged period is conservative as membrane integrity is anticipated to have degraded with time. The results of this study are significant as real pathogen data—in high volume samples and for a 10-year-old flat sheet—has not been reported previously.
Methods
Validation testing of a full-scale MBR facility with Kubota membrane units was performed at the Lake of the Pines Wastewater Treatment Plant (WWTP) in Auburn, California. The WWTP has a maximum monthly flow of 1.4 mgd with an A2O process that includes an MBR, a membrane thickener for sludge management followed by UV disinfection (see Figure 3).
The validation study has included a sampling campaign where high volume sampling (150 to 300 liters) of filtrate has been conducted for the target pathogens specified for evaluation in Tier 2 MBR validation, Cryptosporidium, Giardia and enterovirus. In addition, indicator microorganisms, Clostridium perfringens (for protozoa) and male-specific and somatic coliphages (viruses) have also been monitored. The high-volume sampling has been accomplished with concentration onsite for protozoa using Envirocheck filters and nanoceram filters for virus (Figure 4). These filters are then shipped overnight on ice for extraction and analysis at specialized laboratories.
Results
Results to date have shown virus LRVs that range from 2.0-log for total culturable enterovirus, and >2.3-log for enterovirus analyzed using DNA based techniques.
Protozoa LRVs have ranged from >3.3-log for Cryptosporidium (range >3.3 to >4.6) and >4.2-log for Giardia (range >4.2 to 6.0 LRV). Coliphage LRVs have ranged from >2.8-log for male-specific (range 2.8 to 4.6 LRV) and >1.2-log for somatic (range 1.2 to 5.3 LRV). C. perfringens LRVs have been >2.3-log (range 2.3 to 4.6 LRV) (see Figure 5, page 18).
Although these results are encouraging, the LRVs are still limited by the pathogen influent concentration, even with higher volume sampling. As shown in Figure 6, if the influent concentrations were higher, then the true potential of the MBR could be realized. Follow up sampling with increased volumes is planned at Lake of the Pines WWTP. Additionally, it is apparent that at typical influent concentrations, the MBR is able to provide significant removal of these pathogens to below the limit of detection in the filtrate.
Conclusions
The results of this study suggest that in a well operated system, Kubota flat sheet membranes can continue to achieve significant LRVs beyond the typically expected life. No protozoa LRVs observed were below 3, which confirms that MBRs are appropriate as a replacement for direct MF in membrane-based reuse schemes where over 12-log reduction of virus and 10-log reduction of Cryptosporidium and Giardia are required. In addition, the virus LRV demonstrated in this study is significant, with the conservative indicator organism somatic coliphage always above an LRV of 1.2 and the regulated pathogen, total culturable enterovirus, always above 2. This work contributes significantly to the sparingly available LRV data set for real pathogenic organisms measured across flat sheet MBRs.
Ongoing work
High volume in-line sampling of filtrate suspended solids, particle size distribution analysis and turbidity monitoring are underway with the intention of correlating findings with the established LRV. If successful, these efforts will be one of the first demonstrations of Tier 3 MBR validation globally. In addition, rigorous integrity testing of the used membranes will be compared to unused factory samples. These results are anticipated to provide valuable information on the robust nature of membranes used for wastewater treatment and reuse.